
Therefore, there are various non-equivalent definitions of atomic radius. Chlorine Cl Periodic Table of Elements, Atomic Mass Vector Illustration. 
However, this assumes the atom to exhibit a spherical shape, which is only obeyed for atoms in vacuum or free space. Chlorine atomic structure has atomic number, atomic mass, electron configuration and. The atomic radius of a chemical element is a measure of the distance out to which the electron cloud extends from the nucleus. It must be noted, atoms lack a well-defined outer boundary. The atomic radius of Chlorine atom is 102pm (covalent radius). Note that, each element may contain more isotopes, therefore this resulting atomic mass is calculated from naturally-occuring isotopes and their abundance. The atomic mass is carried by the atomic nucleus, which occupies only about 10 -12 of the total volume of the atom or less, but it contains all the positive charge and at least 99.95% of the total mass of the atom. The atomic mass or relative isotopic mass refers to the mass of a single particle, and therefore is tied to a certain specific isotope of an element. Mass numbers of typical isotopes of Chlorine are 35 37. Isotopes are nuclides that have the same atomic number and are therefore the same element, but differ in the number of neutrons. The difference between the neutron number and the atomic number is known as the neutron excess: D = N – Z = A – 2Z.įor stable elements, there is usually a variety of stable isotopes. Neutron number plus atomic number equals atomic mass number: N+Z=A. The total number of neutrons in the nucleus of an atom is called the neutron number of the atom and is given the symbol N. 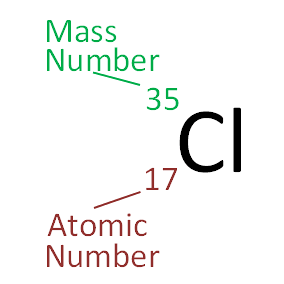
The total electrical charge of the nucleus is therefore +Ze, where e (elementary charge) equals to 1,602 x 10 -19 coulombs. Total number of protons in the nucleus is called the atomic number of the atom and is given the symbol Z. However, three out of four chlorine atoms weigh 35 amu (17 protons and 18 neutrons) and the fourth weighs 37 amu (17 protons and 20 neutrons). For bulk stoichiometric calculations, we are usually determining molar mass, which may also be called standard atomic weight or average atomic mass.Atomic Number – Protons, Electrons and Neutrons in ChlorineĬhlorine is a chemical element with atomic number 17 which means there are 17 protons in its nucleus. This is not the same as molecular mass, which is the mass of a single molecule of well-defined isotopes. This is how to calculate molar mass (average molecular weight), which is based on isotropically weighted averages. The atomic weights used on this site come from NIST, the National Institute of Standards and Technology. Using the chemical formula of the compound and the periodic table of elements, we can add up the atomic weights and calculate molecular weight of the substance. These relative weights computed from the chemical equation are sometimes called equation weights. The formula weight is simply the weight in atomic mass units of all the atoms in a given formula.įormula weights are especially useful in determining the relative weights of reagents and products in a chemical reaction. When calculating molecular weight of a chemical compound, it tells us how many grams are in one mole of that substance. The percentage by weight of any atom or group of atoms in a compound can be computed by dividing the total weight of the atom (or group of atoms) in the formula by the formula weight and multiplying by 100.įinding molar mass starts with units of grams per mole (g/mol). If the formula used in calculating molar mass is the molecular formula, the formula weight computed is the molecular weight. Here is a video which summarizes how to calculate average atomic mass. Calculate the molecular weight of a chemical compound. which gives us the mass shown on the periodic table, 35.5 u. Element: Chlorine Symbol: Cl Atomic Mass: 35.453 of Atoms: 2 Mass Percent: 74.472. This site explains how to find molar mass. A naturally occurring sample of chlorine is 75.78 chlorine-35 and 24.22 chlorine-37, so, to calculate the average mass, we need to do the sum. The reason is that the molar mass of the substance affects the conversion. To complete this calculation, you have to know what substance you are trying to convert. In chemistry, the formula weight is a quantity computed by multiplying the atomic weight (in atomic mass units) of each element in a chemical formula by the number of atoms of that element present in the formula, then adding all of these products together.Ī common request on this site is to convert grams to moles.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
